|
||
pancreas development The pancreas is formed from the ventral and dorsal buds that appear at the foregut-midgut junction at embryonic day (e)9.5 in mice. Over the course of the next 10 days these two clusters of epithelial cells differentiate into the three main cell types in the adult organ: exocrine cells, duct cells, and the hormone-producing islets of Langerhans. The first differentiated islet cells that appear are glucagon positive α-cells at e9.5, β-cells appear a short time later, followed by somatostatin and pancreatic polypeptide producing δ- and PP-cells, respectively. Stages of β-cell genesis The first insulin-expressing cells appear just after bud formation. However, they do not express all the markers of adult β-cells, often co-express glucagon and are likely not physiologically equivalent to mature β-cells; thus, we refer to these atypical cells as primary cells. Between e13.5 and e15.5 there is a rapid, synchronized wave of endocrine differentiation in the pancreas known as the “secondary transition.” During this time, the duct structure of the pancreas forms, the first exocrine cells develop, and β-cell neogenesis peaks. The β-cells present at this time express many of the markers of adult cells, but glucose-stimulated insulin secretion is likely absent. During the “secondary transition”, the endocrine progenitor cells undergo marked changes in both morphology and gene expression. The reasons for these changes are unclear, but because of these differences we refer to these cells as secondary progenitors. As neogenesis wanes at around e18.5, the β-cell proliferation rate increases and peaks around P0 at which time adult islets start to form and insulin is secreted. Subsequent increases in β-cell mass are almost exclusively due to changes in the replication rates of existing cells. |
|
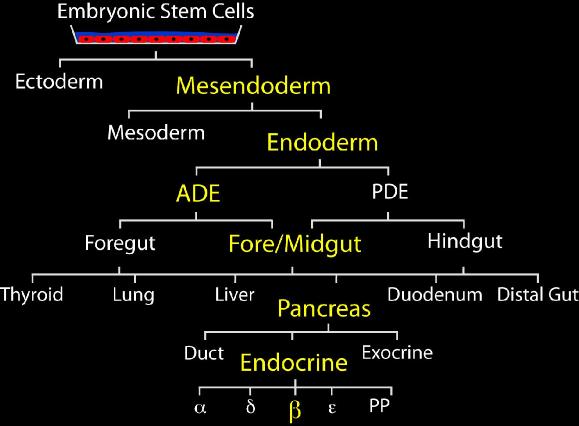 |
Figure 1: Multiple cell fate choices during development from embyonic stem cells to pancreatic beta cells